 
For spot contracts, miners are paid according to the copper price at the time that the smelter/copper refinery makes the sale, not at the copper price on the date of delivery of the concentrate.įor longer-term contracts, pricing is based on an agreed-upon copper price for a future date, typically 90 days from time of delivery to the smelter. Typically, copper concentrate is traded either via spot contracts or under long-term contracts as an intermediate product. All of the risk (and reward) of fluctuating copper prices, then, falls on miners’ shoulders. Smelters generally operate by charging tolls, but they may also sell refined copper metal on behalf of miners. TCs and RCs are levied separately on these metals. Miners may also receive credits for “valuable” minerals, such as precious metals gold and silver. Most smelting companies have strict limitations on permissible concentrations of impurities, and if concentrate producers do not meet these needs, they will be subject to financial penalties. Additionally, penalties may be assessed against copper concentrate according to the level of deleterious elements contained, such as lead or tungsten. Miners indicate copper concentrations, although they may be spot checked by a third party when en route to the refiner. TCs and RCs tend to rise when there is a high availability of copper ore. These charges fluctuate with the market, but are often fixed on an annual basis. 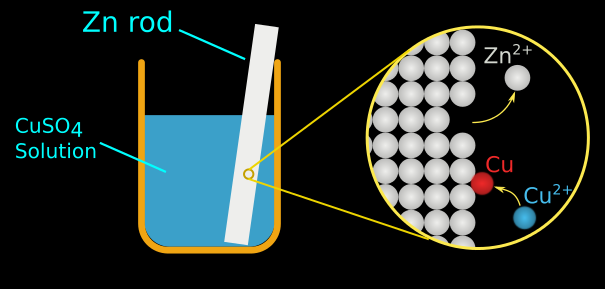
TCs are charged per tonne of concentrate treated, while RCs are charged per pound of metal refined. Selling terms are unique to each smelting company or copper refinery, but in general, the smelter pays the miner approximately 96 percent of the value of the contained copper content in the concentrate, minus treatment charges (TCs) and refining charges (RCs). As mentioned above, copper concentrate is most often refined using equipment at a different location than company mine sites.Ĭoncentrate producers sell a concentrate powder containing 24 to 40 percent copper to copper smelters and refiners. Mining companies may sell copper in concentrate or cathode form. How does refined copper reach the market? 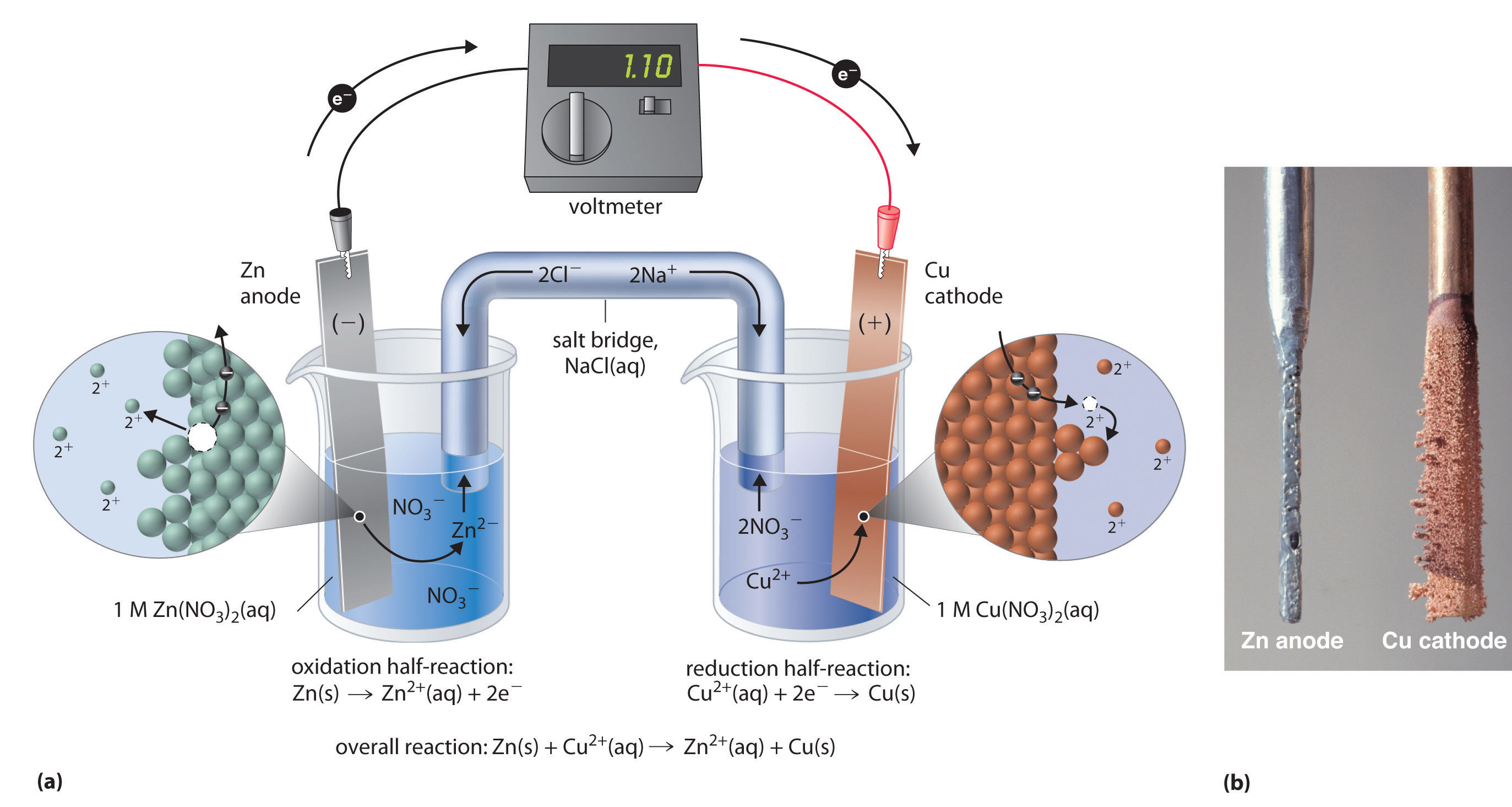
This copper cathode can be sold as is, or can be made into rods or starting sheets for other electrolytic copper cells. This solution goes through the electrowinning process, which plates copper in the solution onto a cathode. Next, sulfuric acid is added to strip the copper from the organic solvent, producing an electrolytic solution. Solvent extraction begins with an organic solvent, which separates copper from impurities and unwanted material. This newer refining technology became widely adopted in the 1980s, and roughly 20 percent of the world’s copper production is now produced via this process. The more common refining method is solvent extraction and electrowinning, or SX/EW. After enough copper has been plated, the copper is then further refined. The least common method is cementation, in which an acidic solution of copper is deposited onto scrap iron in an oxidation-reduction reaction. In hydrometallurgy, copper concentrate undergoes refining via one of a few processes. How is copper extracted by hydrometallurgy? Once charged, the pure copper collects on the cathode and is removed as a 99 percent pure copper product. Finally, to completely deoxidize the copper, either phosphorus is added to it to form P2O5, or the copper is cast into copper anodes and placed in an electrolytic copper cell. In fire refining, air is blown through the copper to oxidize impurities into slag then wood is added to help reduce the oxidized copper through chemical reactions, leaving refined copper behind to be processed into copper cathode. A variety of chemicals are added to the converter, and these react with the copper to form converted copper, called “blister copper.” The blister copper is recovered and is then subjected to a process called fire refining. The slag is discarded and the matte is recovered and moved to a cylindrical vessel called a converter. The matte layer, on the bottom, contains the copper, while the top slag layer contains the impurities. Chemical reactions that occur during the heating process cause the concentrate to segregate into two layers of material: a matte layer and a slag layer. In pyrometallurgy, copper concentrate is dried before being heated in a refining furnace. A short Standard Reduction Potentials table is available from the HandbookMenu, but you may also click the live link to see one.How is copper extracted by pyrometallurgy? These values are usually tabulated in handbooks. Reduction potentials of standard cells have been measured against the SHE or other standards their potentials are measured. Ideally, for every redox couple, there is a reduction potential. \( \newcommand\) ions are less ready to accept electrons than hydrogen ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
